
by wscadmin | Dec 10, 2022 | Blog
Numerous API Intermediate Manufacturers claim that the pharmaceutical industry is complicated and tightly regulated. The raw ingredients required to make completed pharmaceutical goods, such as APIs and drug intermediates, are essential to this sector. The raw...

by wscadmin | Nov 8, 2022 | Blog
The number one priority in the fine chemicals industry is health, safety, and environment (HSE). Still, it is essential to remember that safety has many levels in fine chemical production. First off is the security of the manufacturing method and the workers. Then...
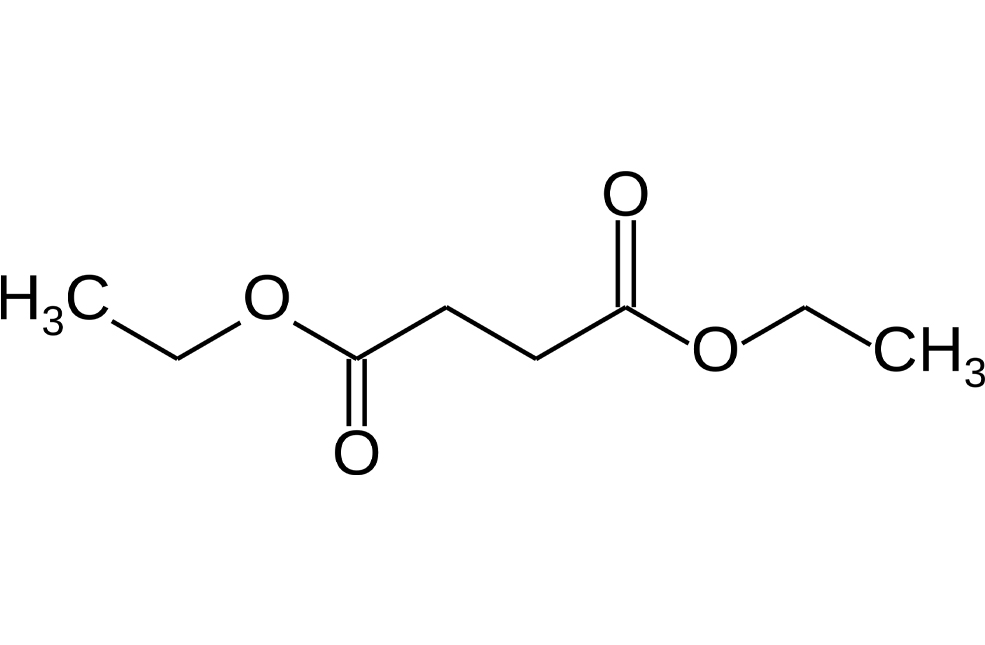
by wscadmin | Oct 8, 2022 | Blog
Introduction Mutagenic impurities, formerly known as genotoxic impurities (GI) or potential genotoxic impurities (PGI), are compounds that can modify DNA and, consequently, can cause cancer. Impurities present in the marketed drug must be evaluated early in the drug...

by wscadmin | Sep 15, 2022 | Blog
Active Pharmaceutical Ingredients are the varied elements of a medical drug. These drugs are produced to Supply the desired impact on the human body which helps in treating Many medical conditions. Multiple app chemical combination mixtures are processed to make an...

by wscadmin | Jul 21, 2022 | Blog
The pharmaceuticals used as raw materials for the creation of bulk drugs are known as bulk drug intermediates. They can also refer to a substance created during the synthesis of an API that needs to go through additional molecular processing or alteration before it is...







Recent Comments